Advances in Delaying T1D Progression
Type 1 diabetes (T1D) is increasingly being treated as a condition requiring early intervention to slow progression, with numerous agents under investigation. To date, teplizumab, an Fc receptor-nonbinding anti-CD3 monoclonal antibody, is the only disease-modifying therapy that is currently approved by the US Food and Drug Administration (FDA). It is approved to delay the onset of stage 3 T1D in patients ≥1 year of age with stage 2 T1D, and is administered as a 14-day outpatient course delivered intravenously with dosing based on body surface area.1
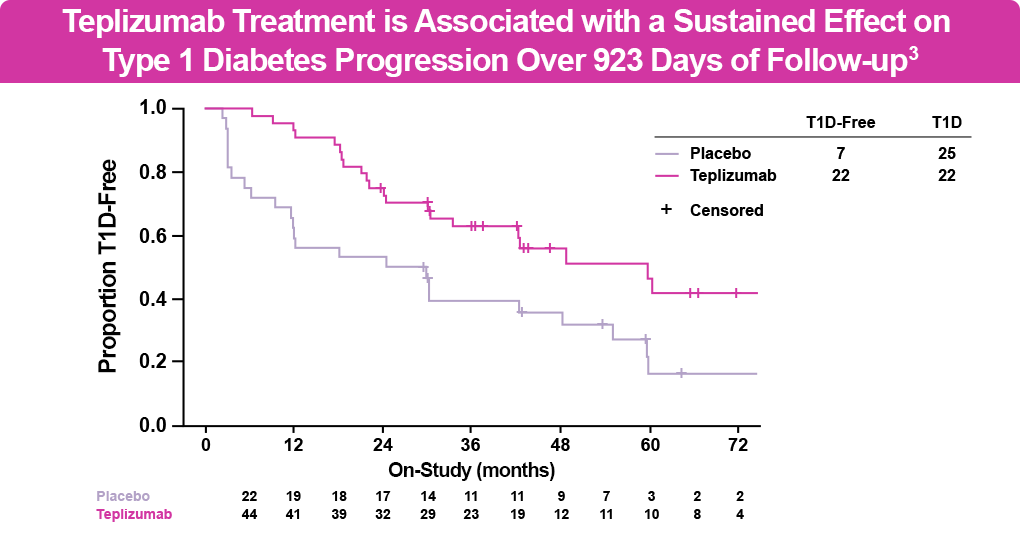
The FDA originally approved teplizumab for this indication in patients ≥8 years of age, based on the phase 2 double-blind At-Risk (TN-10) clinical study of relatives of patients with T1D at high risk for developing clinical disease. In this study, patients received a 14-day course of teplizumab or placebo, with monitoring for progression every 4 months using an oral glucose tolerance test. At 6 years of follow-up, 50% of patients receiving teplizumab were diabetes free vs 22% of those receiving placebo (see figure below). The most common adverse events were rash and transient lymphopenia.2,3 Treatment with teplizumab delayed progression to stage 3 T1D by 32.5 additional months compared with placebo. The median time to diagnosis with teplizumab was 59.6 months vs 27.1 months with placebo, with certain individuals having even longer delays with teplizumab. Additionally, treatment with teplizumab was associated with reversal of C-peptide decline and improved beta cell function compared with placebo.3
In addition, real-world data and patient experience surveys support the application and use of teplizumab to delay T1D progression. In a survey of adults and caregivers of children who were treated with teplizumab to delay the onset of stage 3 T1D, most agreed that they (or the child they care for) were grateful to receive teplizumab and that this treatment would make it easier to manage their T1D. This was the first real-world study to ask about patients’ experiences outside of a clinical trial and can provide additional context for individuals or families who are considering screening for presymptomatic T1D, as well as for clinicians who are considering teplizumab for their patients.4
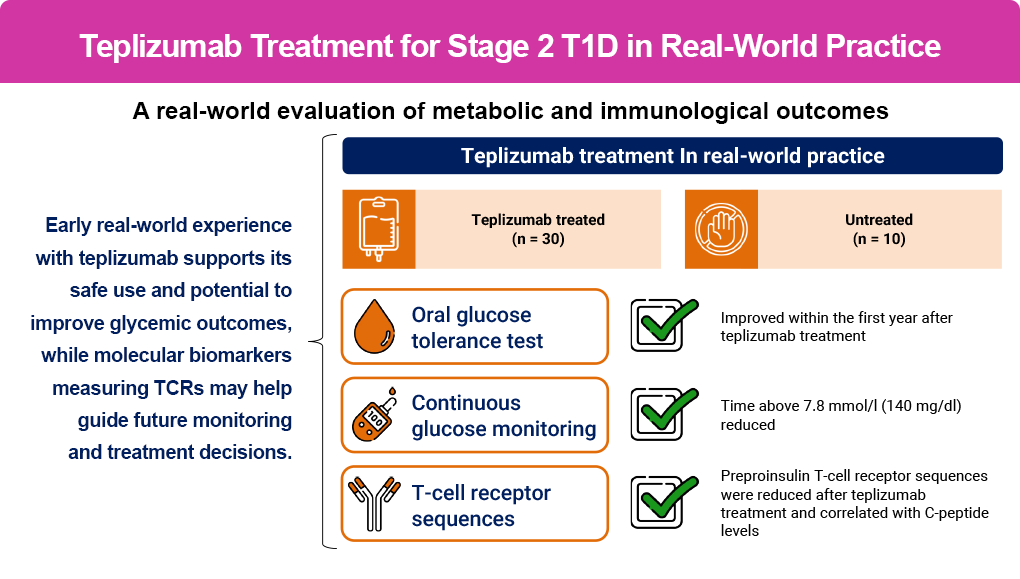
As teplizumab is transitioning to clinical care for this indication, real-world studies will provide additional information beyond clinical trials. A prospective study from the University of Colorado Barbara Davis Center for Diabetes of patients with stage 2 T1D treated with teplizumab was shown to support its safe use and potential to improve glycemic outcomes, as outlined in the figure below.5 Additionally, the study provided some early evidence on potential biomarkers and post-treatment immunologic changes that may guide future monitoring and treatment decisions, linking immunologic response with clinical outcomes and beta cell preservation, although this part is in its very early stages.
Another real-world study looking at global claims data showed that out of 42 patients with stage 2 T1D (11 adults and 31 children and adolescents, aged 8–17 years) treated with teplizumab, only 17% progressed to insulin initiation within approximately the first year, a result consistent with clinical trial data.6
Teplizumab is also being investigated to delay stage 3 progression in patients <8 years of age with stage 2 T1D, as part of the PETITE-T1D study. Preliminary results from the study are shown in the figure below, outlining a similar safety profile that was consistent with other studies as well as demonstrating efficacy for delaying T1D progression in this age group.7 Based on the results of the PETITE-T1D study, in April 2026 teplizumab’s indication for delaying stage 3 T1D was expanded to include patients ≥1 year of age with stage 2 T1D.1
In addition, teplizumab is also being studied in new-onset (stage 3) T1D to preserve beta cells, as outlined in the PROTECT & βETA PRESERVE studies (see more information in the Beta Cell Preservation section). Based on the results of the PROTECT trial, an expanded indication for new-onset (stage 3) T1D is currently under review by the US Food and Drug Administration.
Considerations for Teplizumab Use and Patient Selection
If an individual is confirmed to have stage 2 T1D, and upon appropriate education and shared decision-making about treatment goals with patients and their caregivers, clinicians should consider starting teplizumab to delay progression to stage 3 T1D, or refer eligible patients to specialty care based on the criteria shown below .1 In addition, as outlined in the figure below, there are diagnostic codes specific to presymptomatic T1D, which may help facilitate patient selection.
Teplizumab treatment requires careful patient selection and systematic implementation in dedicated infusion facilities with specially trained staff 8
Before initiating treatment, patients must complete all vaccinations and undergo screening for active infections, including Epstein-Barr virus (EBV), cytomegalovirus (CMV), hepatitis, human immunodeficiency virus (HIV), and tuberculosis (TB). Baseline laboratory parameters required include adequate hemoglobin, platelets, lymphocytes, neutrophils, and liver function.8 Teplizumab treatment may lead to viral reactivation, including EBV and CMV reactivation, and it is important to test for the presence of active EBV or CMV infections before starting treatment, especially in patients who are immunocompromised, as they are at increased risk.1
There are no data on the use of teplizumab during pregnancy; thus, its use is not recommended for more than 30 days prior to planned pregnancy or during pregnancy. Individuals of reproductive age should also have a negative pregnancy test prior to administration.8
The treatment consists of 14 consecutive daily intravenous (IV) infusions (either via peripheral or central venous access) with an escalating dose schedule, beginning at 65 μg/m² on Day 1 and increasing to a maintenance dose of 1,030 μg/m² by Day 5. The protocol escalates the dose over the first 5 days because cytokine release syndrome (CRS) is most common early; but it can largely be prevented by pretreatment with an antipyretic and antihistamine given at least 30 minutes before infusion (max dose/age).
Each infusion takes at least 30 minutes for patients ≥8 years of age, and at least 2 hours for patients 1 to 7 years of age.1 However, additional time should be factored in on infusion days for pre-infusion assessment and pre-medications, potential lab work, and post-infusion monitoring.8 For detailed information and additional resources on the administration of teplizumab, you can visit the Infusion portal of the DETECT-T1D website.
Post-treatment management involves monitoring for progression to stage 3 T1D every 6 months, typically through OGTT or continuous glucose monitoring.8 A summary of these recommendations is shown in the table below, which are part of the 2024 Pediatric Endocrine Society (PES) guidance on teplizumab treatment.8
Teplizumab is also being investigated in the phase 4 PETITE-T1D trial in patients <8 years of age with stage 2 T1D (NCT05757713) and was also investigated in a phase 2 extension (NCT04270942) of the AT-Risk (TN-10) trial.
Other Emerging Disease-Modifying Therapies for T1D
Several other investigational agents are in development. To date, most have been tested in new-onset T1D, some with mixed results; additional studies are ongoing. These studies include additional approaches to modulating or depleting effector T cells, decreasing B cells and antigen presentation, as well as anti-inflammatory and beta cell protective agents. Some of the key investigational agents are discussed below.
In addition, real-world data and patient experience surveys support the application and use of teplizumab to delay T1D progression. In a survey of adults and caregivers of children who were treated with teplizumab to delay the onset of stage 3 T1D, most agreed that they (or the child they care for) were grateful to receive teplizumab and that this treatment would make it easier to manage their T1D. This was the first real-world study to ask about patients’ experiences outside of a clinical trial and can provide additional context for individuals or families who are considering screening for presymptomatic T1D, as well as for clinicians who are considering teplizumab for their patients.4
As teplizumab is transitioning to clinical care for this indication, real-world studies will provide additional information beyond clinical trials. A prospective study from the University of Colorado Barbara Davis Center for Diabetes of patients with stage 2 T1D treated with teplizumab was shown to support its safe use and potential to improve glycemic outcomes, as outlined in the figure below.5 Additionally, the study provided some early evidence on potential biomarkers and post-treatment immunologic changes that may guide future monitoring and treatment decisions, linking immunologic response with clinical outcomes and beta cell preservation, although this part is in its very early stages.
Another real-world study looking at global claims data showed that out of 42 patients with stage 2 T1D (11 adults and 31 children and adolescents, aged 8–17 years) treated with teplizumab, only 17% progressed to insulin initiation within approximately the first year, a result consistent with clinical trial data.6
Teplizumab is also being investigated to delay stage 3 progression in patients <8 years of age with stage 2 T1D, as part of the PETITE-T1D study. Preliminary results from the study are shown in the figure below, outlining a similar safety profile that was consistent with other studies as well as demonstrating efficacy for delaying T1D progression in this age group.7 Based on the results of the PETITE-T1D study, teplizumab is under US Food and Drug Administration (FDA) review for a potential expanded indication to delay progression to stage 3 in patients <8 years of age.
Besides teplizumab, several other investigational agents are in development. To date, most have been tested in new-onset T1D—some with mixed results. Additional studies are ongoing. These studies include additional approaches to modulating or depleting effector T cells, decreasing B cells and antigen presentation, as well as anti-inflammatory and beta cell protective agents. More information about ongoing trials can be found in the Reading & Resources section.
References
- Teplizumab (Tzield®) Prescribing information 2026. Sanofi. https://products.sanofi.us/tzield/tzield.pdf
- Herold KC, Bundy BN, Long SA, et al. An anti-CD3 antibody, teplizumab, in relatives at risk for type 1 diabetes. N Engl J Med. 2019;381:603-613.
- Sims EK, Bundy BN, Stier K, et al. Teplizumab improves and stabilizes beta cell function in antibody-positive high-risk individuals. Sci Transl Med. 2021;13:eabc8980.
- O’Donnell HK, Simmons KM, Gitelman SE, et al. Real-world experience with teplizumab in stage 2 type 1 diabetes: a plain language summary. Ther Adv Endocrinol Metab. 2026;17:20420188261417154.
- Karakus KE, Chesshir L, Walker S, et al. Teplizumab treatment for stage 2 type 1 diabetes: a real-world evaluation of metabolic and immunological outcomes. Diabetologia. 2026;69:1164-1175.
- Mahesh S, Anson M, Malik RA, et al. Real-world evaluation of teplizumab in type 1 diabetes: progression to insulin requirement. Diabetes Obs Metab. 2026;28:2425-2430.
- Gitelman SE, Simmons K, Sherr JL, et al. Safety and pharmacokinetics of teplizumab in children less than 8 years of age with stage 2 type 1 diabetes. Diabetologia. 2026;69:330-342.
- Mehta S, Ryabets-Lienhard A, Patel N, et al. Pediatric Endocrine Society statement on considerations for use of teplizumab (Tzield™) in clinical practice. Horm Res Paediatr. 2024; 98:597-608.